In my blog as well as presenting some serious science, I try and bring you interesting and quirky items I come across. Today’s post falls into the ‘interesting and quirky’ and is something I’ve been able to find out almost nothing about online. It is a Carl Zeiss Quarz Silberfilter (Quartz Silver filter) which I bought on ebay for the princely sum of £10 including delivery.
So what is this thing? It’s an optical filter with the markings “Carl Zeiss Jena” and “Quarz-Silberfilter x37” on it. It is a push fit filter with a diameter of 37mm (presumably that led to the x37 on the name). The optical part of the filter just looks like a mirror on both sides, and the mirror finish is sandwiched in the middle between two pieces of quartz, I am assuming to protect the mirror coating as much as possible. Overall, it is about 5mm thick and is held in with a threaded ring. Here’s some photos of it.




The filter does show some degradation around the edges, but this looks to be minor.
What is this filter, and why did it pique my interest? Often (but not always) Quartz or as it is spelt here ‘Quarz’ can mean that it is for a UV application. This got me wondering whether it was a filter designed to block visible light and let through part of the UV spectrum. Silver has an interesting property in the UV that it reflects most wavelengths, apart from around 310-320nm where it absorbs quite strongly. I have shown this effect before by imaging silver foil at different wavelengths in the UV here. After an online search I found one mention of Quartz Silver filter in translation of a 1920 document “The Palimpsest Photography
(Photography of the etched writings)” which talked about them being used to isolate the 313nm line. Aha I thought, it is using silver’s optical properties to not reflect light around 320nm-ish while reflecting other wavelengths. Digging bit more, I came across an article from 1911 in the Smithosonian Institution Annual report called “Recent experiments with invisible light” which talked about silvering of quartz lenses to isolate parts of the UV spectrum for imaging. In that article it states that “If we deposit chemically a thin film of metallic silver on the surface of a quartz lens, a certain amount of ultra-violet radiation between 3000 and 3200 [Angstroms, or 300-320nm] is able to struggle through and form an image on the plate”.
So, it seemed as though this is a filter for imaging in the low 300nm region, and that it would have low transmission even in the region of interest (given the light ‘struggles’ through. I love old science journals and their terminology). I was not able to find a transmission spectrum for the Zeiss filter online, so I measured it using my Ocean Optics FX spectrometer, the results of which are shown below.
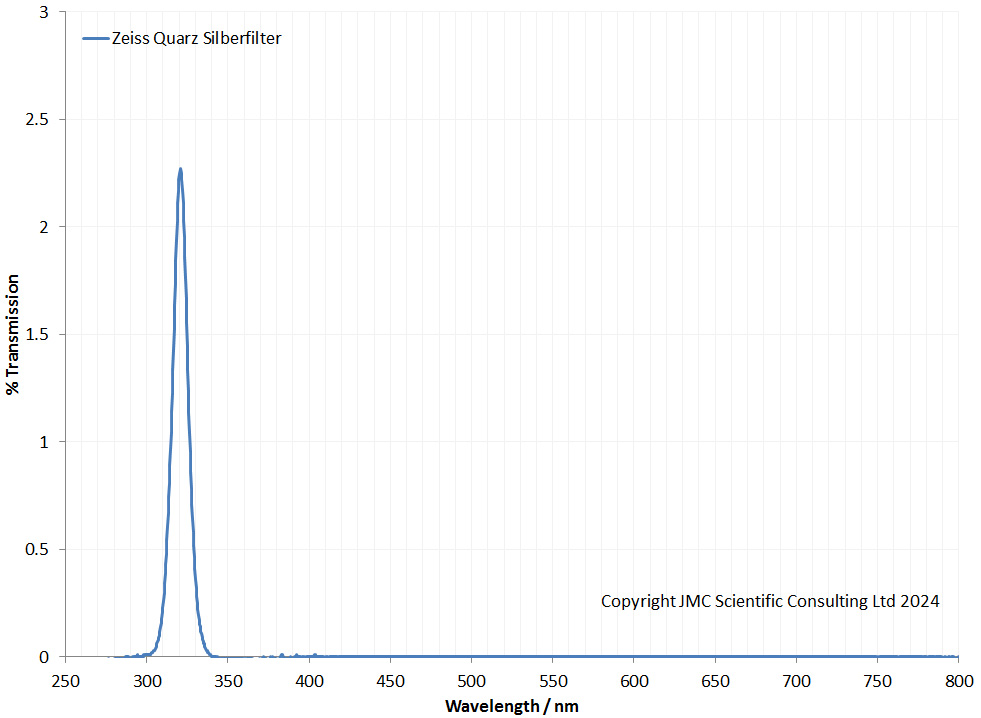
Blimey they weren’t joking about light struggling through. It does indeed isolate the light around the 320nm region. However max transmission was only just over 2%. But it was very effective at blocking unwanted wavelengths. I guess if you had a nice powerful UV source (such a mercury xenon source) and you wanted to isolate the 313nm line, this type of filter could be used for that. Photographic paper could be made to be reasonably UV sensitive unlike normal digital camera sensors, so perhaps this was ‘good enough’ for imaging purposes.
As an aside, a Quarz Silberfilter is also mentioned in the brochure for the Zeiss 60mm f4 UV-Objectiv (a camera lens I have written about here). However this one is not designed to fit directly on to that lens – the filter ring on that lens is threaded and a larger diameter than this filter. So perhaps Zeiss designed one specifically for that lens as well.
Is it still useful? Probably not, given the low transmission. Nowadays dichroic filters can be made with relatively high transmission and good blocking of out of wavelength bands. However it is an interesting scientific curiosity, and a throwback to an early bandpass filter design for looking at the UV spectrum. Am I gutted I spent £10 on it? No, although I wouldn’t have wanted to spend any more on it. As I say, it’s a quirky piece of scientific history.
As always, thanks for reading, and if you’d like to know more about my work, I can be reached here.